Sequence
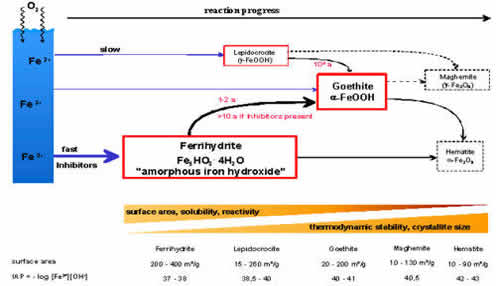
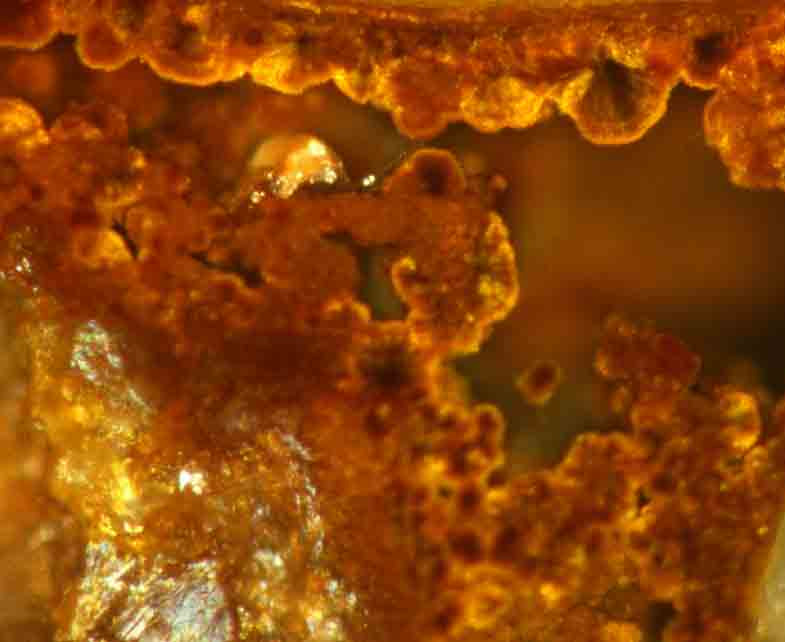
Microorganisms living in the soil and in aquifers obtain their vital energy from the oxidisation of bivalent iron (iron II) in trivalent iron (iron III). The insoluble iron III originates from the metabolism of the bacteria and precipitates as hydrous and voluminous iron oxide forming well incrustations. Metastable phases start to crystalise and form soft mineral ferrihydrite.

Sediment from Leptothrix ochracea
Spirals:
Sediment from Gallionella ferruginea (Banfield & Zhang 2001)

Crystallisation and recrystallization of iron oxides
Soft ferrihydrate and lepidocrocite with high surface energy change gradually into thermodynamically more stable goethite with much less surface energy. At the same time the insolubility and the crystal size increase dramatically.
Precipitation
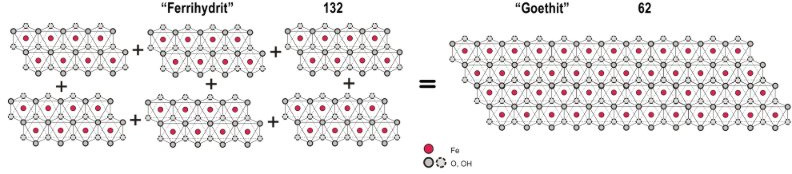
The surface area, the solubility and the reactivity of iron oxides decrease during the ageing process. Younger incrustations shield the older ones so that annual rings can be seen in a microscope.
This explains why it is much more difficult to remove and dissolve older incrustations. The rehabilitation should be carried out when the incrustations are young and consist of easily soluble mineral phases.









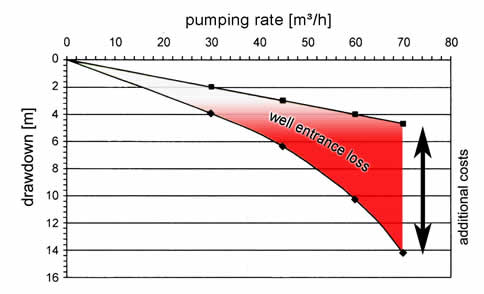 » Calculate here
» Calculate here » Have fun!
» Have fun!